Brangwynne CP, Eckmann CR, Courson DS, Rybarska A, Hoege C, Gharakhani J, et al. Germline P granules are liquid droplets that localize by controlled dissolution/condensation. Science. 2009;324:1729–32.
Banani SF, Lee HO, Hyman AA, Rosen MK. Biomolecular condensates: organizers of cellular biochemistry. Nat Rev Mol Cell Biol. 2017;18:285–98.
Conti BA, Oppikofer M. Biomolecular condensates: new opportunities for drug discovery and RNA therapeutics. Trends Pharmacol Sci. 2022;43:820–37.
Mitrea DM, Mittasch M, Gomes BF, Klein IA, Murcko MA. Modulating biomolecular condensates: a novel approach to drug discovery. Nat Rev Drug Discov. 2022;21:841–62.
Boija A, Klein IA, Young RA. Biomolecular condensates and cancer. Cancer Cell. 2021;39:174–92.
Silva JL, Foguel D, Ferreira VF, Vieira T, Marques MA, Ferretti GDS, et al. Targeting biomolecular condensation and protein aggregation against cancer. Chem Rev. 2023;123:9094–138.
Ahn JH, Davis ES, Daugird TA, Zhao S, Quiroga IY, Uryu H, et al. Phase separation drives aberrant chromatin looping and cancer development. Nature. 2021;595:591–5.
Chong S, Graham TGW, Dugast-Darzacq C, Dailey GM, Darzacq X, Tjian R. Tuning levels of low-complexity domain interactions to modulate endogenous oncogenic transcription. Mol Cell. 2022;82:2084–97.e5.
Mehta S, Zhang J. Liquid-liquid phase separation drives cellular function and dysfunction in cancer. Nat Rev Cancer. 2022;22:239–52.
Zhang XW, Yan XJ, Zhou ZR, Yang FF, Wu ZY, Sun HB, et al. Arsenic trioxide controls the fate of the PML-RARalpha oncoprotein by directly binding PML. Science. 2010;328:240–3.
Li P, Banjade S, Cheng HC, Kim S, Chen B, Guo L, et al. Phase transitions in the assembly of multivalent signalling proteins. Nature. 2012;483:336–40.
Gouveia B, Kim Y, Shaevitz JW, Petry S, Stone HA, Brangwynne CP. Capillary forces generated by biomolecular condensates. Nature. 2022;609:255–64.
Yasuda S, Tsuchiya H, Kaiho A, Guo Q, Ikeuchi K, Endo A, et al. Stress- and ubiquitylation-dependent phase separation of the proteasome. Nature. 2020;578:296–300.
Weber SC, Brangwynne CP. Inverse size scaling of the nucleolus by a concentration-dependent phase transition. Curr Biol. 2015;25:641–6.
Sieme D, Engelke M, Rezaei-Ghaleh N, Becker S, Wienands J, Griesinger C. Autoinhibition in the signal transducer CIN85 modulates B cell activation. J Am Chem Soc. 2024;146:399–409.
Fabrini G, Farag N, Nuccio SP, Li S, Stewart JM, Tang AA, et al. Co-transcriptional production of programmable RNA condensates and synthetic organelles. Nat Nanotechnol. 2024;19:1665–73.
Millar SR, Huang JQ, Schreiber KJ, Tsai YC, Won J, Zhang J, et al. A new phase of networking: the molecular composition and regulatory dynamics of mammalian stress granules. Chem Rev. 2023;123:9036–64.
Chen X, Fansler MM, Janjoš U, Ule J, Mayr C. The FXR1 network acts as a signaling scaffold for actomyosin remodeling. Cell. 2024;187:5048–63.e25.
Holehouse AS, Kragelund BB. The molecular basis for cellular function of intrinsically disordered protein regions. Nat Rev Mol Cell Biol. 2024;25:187–211.
Na Z, Luo Y, Cui DS, Khitun A, Smelyansky S, Loria JP, et al. Phosphorylation of a human microprotein promotes dissociation of biomolecular condensates. J Am Chem Soc. 2021;143:12675–87.
Luo XJ, He MM, Liu J, Zheng JB, Wu QN, Chen YX, et al. LncRNA TMPO-AS1 promotes esophageal squamous cell carcinoma progression by forming biomolecular condensates with FUS and p300 to regulate TMPO transcription. Exp Mol Med. 2022;54:834–47.
Kang H, Xu T. N6-methyladenosine RNA methylation modulates liquid‒liquid phase separation in plants. Plant Cell. 2023;35:3205–13.
Takakuwa H, Yamazaki T, Souquere S, Adachi S, Yoshino H, Fujiwara N, et al. Shell protein composition specified by the lncRNA NEAT1 domains dictates the formation of paraspeckles as distinct membraneless organelles. Nat Cell Biol. 2023;25:1664–75.
Markaki Y, Gan Chong J, Wang Y, Jacobson EC, Luong C, Tan SYX, et al. Xist nucleates local protein gradients to propagate silencing across the X chromosome. Cell. 2021;184:6174–92.e32.
Saurabh S, Chong TN, Bayas C, Dahlberg PD, Cartwright HN, Moerner WE, et al. ATP-responsive biomolecular condensates tune bacterial kinase signaling. Sci Adv. 2022;8:eabm6570.
Zhu L, Pan Y, Hua Z, Liu Y, Zhang X. Ionic effect on the microenvironment of biomolecular condensates. J Am Chem Soc. 2024;146:14307–17.
Martínez-Cenalmor P, Martínez AE, Moneo-Corcuera D, González-Jiménez P, Pérez-Sala D. Oxidative stress elicits the remodeling of vimentin filaments into biomolecular condensates. Redox Biol. 2024;75:103282.
Shin Y, Brangwynne CP. Liquid phase condensation in cell physiology and disease. Science. 2017;357:eaaf4382.
Shapiro DM, Deshpande S, Eghtesadi SA, Zhong M, Fontes CM, Fiflis D, et al. Synthetic biomolecular condensates enhance translation from a target mRNA in living cells. Nat Chem. 2025;17:448–56.
Kuang J, Li P, Zhai Z, Fan Y, Xu H, Zhao C, et al. Exclusion of HDAC1/2 complexes by oncogenic nuclear condensates. Mol Cancer. 2024;23:85.
Durall RT, Huang J, Wojenski L, Huang Y, Gokhale PC, Leeper BA, et al. The BRD4-NUT fusion alone drives malignant transformation of NUT carcinoma. Cancer Res. 2023;83:3846–60.
Zheng H, Li S, Hsu P, Qu CK. Induction of a tumor-associated activating mutation in protein tyrosine phosphatase Ptpn11 (Shp2) enhances mitochondrial metabolism, leading to oxidative stress and senescence. J Biol Chem. 2013;288:25727–38.
Rowling PJE, Murton BL, Du Z, Itzhaki LS. Multivalent interaction of beta-catenin with its intrinsically disordered binding partner adenomatous polyposis coli. Front Mol Biosci. 2022;9:896493.
Jevtic Z, Allram M, Grebien F, Schwaller J. Biomolecular condensates in myeloid leukemia: what do they tell us?. Hemasphere. 2023;7:e923 Jul.
Sabari BR, Dall’Agnese A, Boija A, Klein IA, Coffey EL, Shrinivas K, et al. Coactivator condensation at super-enhancers links phase separation and gene control. Science. 2018;361:eaar3958.
Crump NT, Ballabio E, Godfrey L, Thorne R, Repapi E, Kerry J, et al. BET inhibition disrupts transcription but retains enhancer-promoter contact. Nat Commun. 2021;12:223.
Yu D, Liang Y, Kim C, Jaganathan A, Ji D, Han X, et al. Structural mechanism of BRD4-NUT and p300 bipartite interaction in propagating aberrant gene transcription in chromatin in NUT carcinoma. Nat Commun. 2023;14:378.
Kiang KM, Ahad L, Zhong X, Lu QR. Biomolecular condensates: hubs of Hippo-YAP/TAZ signaling in cancer. Trends Cell Biol. 2024;34:566–77.
Gemeinhardt TM, Regy RM, Phan TM, Pal N, Sharma J, Senkovich O, et al. A disordered linker in the Polycomb protein Polyhomeotic tunes phase separation and oligomerization. Mol Cell. 2025;85:2128–46.e15.
Kovar H. Dr. Jekyll and Mr. Hyde: the two faces of the FUS/EWS/TAF15 protein family. Sarcoma. 2011;2011:837474.
Lee AY, Chiang CM. Chromatin adaptor Brd4 modulates E2 transcription activity and protein stability. J Biol Chem. 2009;284:2778–86.
Shi B, Li W, Song Y, Wang Z, Ju R, Ulman A, et al. UTX condensation underlies its tumour-suppressive activity. Nature. 2021;597:726–31.
Dai Z, Li G, Chen Q, Yang X. Ser392 phosphorylation modulated a switch between p53 and transcriptional condensates. Biochim Biophys Acta Gene Regul Mech. 2022;1865:194827.
Xie F, Zhou X, Ran Y, Li R, Zou J, Wan S, et al. Targeting FOXM1 condensates reduces breast tumour growth and metastasis. Nature. 2025;638:1112–21.
Lu B, Qiu R, Wei J, Wang L, Zhang Q, Li M, et al. Phase separation of phospho-HDAC6 drives aberrant chromatin architecture in triple-negative breast cancer. Nat Cancer. 2024;5:1622–40.
Zong Z, Xie F, Wang S, Wu X, Zhang Z, Yang B, et al. Alanyl-tRNA synthetase, AARS1, is a lactate sensor and lactyltransferase that lactylates p53 and contributes to tumorigenesis. Cell. 2024;187:2375–92.e33.
Li H, Liu C, Li R, Zhou L, Ran Y, Yang Q, et al. AARS1 and AARS2 sense L-lactate to regulate cGAS as global lysine lactyltransferases. Nature. 2024;634:1229–37.
Patil A, Strom AR, Paulo JA, Collings CK, Ruff KM, Shinn MK, et al. A disordered region controls cBAF activity via condensation and partner recruitment. Cell. 2023;186:4936–55.e26.
Weinert BT, Narita T, Satpathy S, Srinivasan B, Hansen BK, Schölz C, et al. Time-resolved analysis reveals rapid dynamics and broad scope of the CBP/p300 acetylome. Cell. 2018;174:231–44.e12.
Wei H, Zheng H, Wang S, Yang Y, Zhao R, Gu A, et al. Targeting redox-sensitive MBD2-NuRD condensate in cancer cells. Nat Cell Biol. 2025;27:801–16.
Loehr J, Kougnassoukou Tchara PE, Gonthier K, Noufi C, Linteau N, Audet-Walsh É, et al. A nutrient-based cellular model to characterize acetylation-dependent protein-protein interactions. Front Mol Biosci. 2022;9:831758.
Sabò A, Amati B. Genome recognition by MYC. Cold Spring Harb Perspect Med. 2014;4:a014191.
Schütz S, Bergsdorf C, Hänni-Holzinger S, Lingel A, Renatus M, Gossert AD, et al. Intrinsically disordered regions in the transcription factor MYC:MAX modulate DNA binding via intramolecular interactions. Biochemistry. 2024;63:498–511.
Williams ET, Schiefelbein K, Schuster M, Ahmed IMM, De Vries M, Beveridge R, et al. Rapid flow-based synthesis of post-translationally modified peptides and proteins: a case study on MYC’s transactivation domain. Chem Sci. 2024;15:8756–65.
Jiang J, Wang J, Yue M, Cai X, Wang T, Wu C, et al. Direct phosphorylation and stabilization of MYC by aurora B kinase promote T-cell leukemogenesis. Cancer Cell. 2020;37:200–15.e5.
Nguyen NTB, Gevers S, Kok RNU, Burgering LM, Neikes H, Akkerman N, et al. Lactate controls cancer stemness and plasticity through epigenetic regulation. Cell Metab. 2025;37:903–19.e10.
Meyer K, Yserentant K, Cheloor-Kovilakam R, Ruff KM, Chung CI, Shu X, et al. YAP charge patterning mediates signal integration through transcriptional co-condensates. Nat Commun. 2025;16:7454.
Hu X, Wu X, Berry K, Zhao C, Xin D, Ogurek S, et al. Nuclear condensates of YAP fusion proteins alter transcription to drive ependymoma tumourigenesis. Nat Cell Biol. 2023;25:323–36.
Kalita B, Martinez-Cebrian G, McEvoy J, Allensworth M, Knight M, Magli A, et al. PAX translocations remodel mitochondrial metabolism through altered leucine usage in rhabdomyosarcoma. Cell. 2025;188:2757–77.e22.
Li W, Shi X, Tan C, Jiang Z, Li M, Ji Z, et al. Plasma membrane-associated ARAF condensates fuel RAS-related cancer drug resistance. Nat Chem Biol. 2025;21:1226–37.
Lacroix E, Momchilova EA, Chandhok S, Padavu M, Zapf R, Audas TE. PI3K/AKT signaling mediates stress-inducible amyloid formation through c-Myc. Cell Rep. 2025;44:115617.
Jia Y, Jia R, Dai Z, Zhou J, Ruan J, Chng W, et al. Stress granules in cancer: adaptive dynamics and therapeutic implications. iScience. 2024;27:110359.
Yang P, Mathieu C, Kolaitis RM, Zhang P, Messing J, Yurtsever U, et al. G3BP1 Is a tunable switch that triggers phase separation to assemble stress granules. Cell. 2020;181:325–45.e28.
Huang C, Huang J, Lu R, Yu AJ, Yu AS, Cao Y, et al. PABPC1 SUMOylation enhances cell survival by promoting mitophagy through stabilizing U-rich mRNAs within stress granules. Nat Commun. 2025;16:7308.
Lee AK, Klein J, Fon Tacer K, Lord T, Oatley MJ, Oatley JM, et al. Translational repression of G3BP in cancer and germ cells suppresses stress granules and enhances stress tolerance. Mol Cell. 2020;79:645–59.e9.
Buchan JR, Kolaitis RM, Taylor JP, Parker R. Eukaryotic stress granules are cleared by autophagy and Cdc48/VCP function. Cell. 2013;153:1461–74.
Woldemichael GM, Turbyville TJ, Vasselli JR, Linehan WM, McMahon JB. Lack of a functional VHL gene product sensitizes renal cell carcinoma cells to the apoptotic effects of the protein synthesis inhibitor verrucarin A. Neoplasia. 2012;14:771–7.
Liu Y, Li Y, Zhang P. Stress granules and organelles: coordinating cellular responses in health and disease. Protein Cell. 2025;16:418–38.
Cao Y, Lu C, Beeraka NM, Efetov S, Enikeev M, Fu Y, et al. Exploring the relationship between anastasis and mitochondrial ROS-mediated ferroptosis in metastatic chemoresistant cancers: a call for investigation. Front Immunol. 2024;15:1428920.
Li M, Li M, Xia Y, Li G, Su X, Wang D, et al. HDAC1/3-dependent moderate liquid-liquid phase separation of YY1 promotes METTL3 expression and AML cell proliferation. Cell Death Dis. 2022;13:992.
Liu Y, Li Q, Song L, Gong C, Tang S, Budinich KA, et al. Condensate-promoting ENL mutation drives tumorigenesis in vivo through dynamic regulation of histone modifications and gene expression. Cancer Discov. 2024;14:1522–46.
Seton-Rogers S. SPOP mutations disrupt phase separation. Nat Rev Cancer. 2018;18:667.
Deng Z, Pu L, Deng K, Liu W, Zhang J, Zhang L, et al. Targeting TFAP2β condensation suppresses the development of esophageal squamous cell carcinoma. Cell. 2025;189:1–19.
Cairo LV, Hong X, Müller MBD, Yuste-Checa P, Jagadeesan C, Bracher A, et al. Stress-dependent condensate formation regulated by the ubiquitin-related modifier Urm1. Cell. 2024;187:4656–73.e28.
Sun Y, Zhang S, Hu J, Tao Y, Xia W, Gu J, et al. Molecular structure of an amyloid fibril formed by FUS low-complexity domain. iScience. 2022;25:103701.
Aulas A, Lyons SM, Fay MM, Anderson P, Ivanov P. Nitric oxide triggers the assembly of “type II” stress granules linked to decreased cell viability. Cell Death Dis. 2018;9:1129.
Lu S, Hu J, Arogundade OA, Goginashvili A, Vazquez-Sanchez S, Diedrich JK, et al. Heat-shock chaperone HSPB1 regulates cytoplasmic TDP-43 phase separation and liquid-to-gel transition. Nat Cell Biol. 2022;24:1378–93.
Wei Y, Li D, Yang R, Liu Y, Luo X, Zhao W, et al. TIA1-mediated stress granules promote neurodegeneration by sequestering HSP70 mRNA in C9orf72 mice. Brain. 2025;148:4482–94.
Lennicke C, Cochemé HM. Redox metabolism: ROS as specific molecular regulators of cell signaling and function. Mol Cell. 2021;81:3691–707.
Yang Y, Willis TL, Button RW, Strang CJ, Fu Y, Wen X, et al. Cytoplasmic DAXX drives SQSTM1/p62 phase condensation to activate Nrf2-mediated stress response. Nat Commun. 2019;10:3759.
Dong F, Cheng X, Wan J, Li Q, Du W, Li W, et al. Transcriptional condensates enrich phosphorylated PRMT2 to stimulate H3R8me2a deposition and hypoxic response in glioblastoma. Sci China Life Sci. 2025;69:224–38.
Fang W, Liao C, Shi R, Simon JM, Ptacek TS, Zurlo G, et al. ZHX2 promotes HIF1α oncogenic signaling in triple-negative breast cancer. Elife. 2021;10:e70412.
Gao C, Gao A, Jiang Y, Gao R, Guo Y, Peng Z, et al. Hypoxia-induced phase separation of ZHX2 alters chromatin looping to drive cancer metastasis. Mol Cell. 2025;85:1525–42.e10.
Fuller GG, Kim JK. Compartmentalization and metabolic regulation of glycolysis. J Cell Sci. 2021;134:jcs258469.
Ferrari M, Rawal CC, Lodovichi S, Vietri MY, Pellicioli A. Rad9/53BP1 promotes DNA repair via crossover recombination by limiting the Sgs1 and Mph1 helicases. Nat Commun. 2020;11:3181.
Bigot N, Day M, Baldock RA, Watts FZ, Oliver AW, Pearl LH. Phosphorylation-mediated interactions with TOPBP1 couple 53BP1 and 9-1-1 to control the G1 DNA damage checkpoint. Elife. 2019;8:e44353.
Wang H, Guo W, Mitra J, Hegde PM, Vandoorne T, Eckelmann BJ, et al. Mutant FUS causes DNA ligation defects to inhibit oxidative damage repair in amyotrophic lateral sclerosis. Nat Commun. 2018;9:3683.
Luwang JW, Nair AR, Natesh R. Stability of p53 oligomers: tetramerization of p53 impinges on its stability. Biochimie. 2021;189:99–107.
Petronilho EC, de Andrade GC, de Sousa GDS, Almeida FP, Mota MF, Gomes A, et al. Oncogenic p53 triggers amyloid aggregation of p63 and p73 liquid droplets. Commun Chem. 2024;7:207.
Meng F, Li H, Huang Y, Wang C, Liu Y, Zhang C, et al. RIOK1 phase separation restricts PTEN translation via stress granules activating tumor growth in hepatocellular carcinoma. Erratum : Nat Cancer. 2025;6:1612 Nat Cancer. 2025 Jul;6(7):1223-41Sep.
Wang P, Benhenda S, Wu H, Lallemand-Breitenbach V, Zhen T, Jollivet F, et al. RING tetramerization is required for nuclear body biogenesis and PML sumoylation. Nat Commun. 2018;9:1277.
Cui J, Germer K, Wu T, Wang J, Luo J, Wang SC, et al. Cross-talk between HER2 and MED1 regulates tamoxifen resistance of human breast cancer cells. Cancer Res. 2012;72:5625–34.
Zhang Q, Qiu R, Lu B, Wang J, Cao J, Zhu H, et al. Deacetylase-independent HDAC1 condensation defines temozolomide response in glioblastoma. Nat Chem Biol. Forthcoming 2026. https://doi.org/10.1038/s41589-025-02123-8.
Xie J, He H, Kong W, Li Z, Gao Z, Xie D, et al. Targeting androgen receptor phase separation to overcome antiandrogen resistance. Nat Chem Biol. 2022;18:1341–50.
Yu F, Yu C, Li F, Zuo Y, Wang Y, Yao L, et al. Wnt/β-catenin signaling in cancers and targeted therapies. Signal Transduct Target Ther. 2021;6:307.
Qamar S, Wang G, Randle SJ, Ruggeri FS, Varela JA, Lin JQ, et al. FUS phase separation is modulated by a molecular chaperone and methylation of arginine cation-π interactions. Cell. 2018;173:720–34.e15.
Wall ML, Lewis SM. Methylarginines within the RGG-Motif region of hnRNP A1 affect its IRES trans-acting factor activity and are required for hnRNP A1 stress granule localization and formation. J Mol Biol. 2017;429:295–307.
Anderson EN, Morera AA, Kour S, Cherry JD, Ramesh N, Gleixner A, et al. Traumatic injury compromises nucleocytoplasmic transport and leads to TDP-43 pathology. Elife. 2021;10:e67587.
Liu Z, Zhang S, Gu J, Tong Y, Li Y, Gui X, et al. Hsp27 chaperones FUS phase separation under the modulation of stress-induced phosphorylation. Nat Struct Mol Biol. 2020;27:363–72.
Meng F, Yu Z, Zhang D, Chen S, Guan H, Zhou R, et al. Induced phase separation of mutant NF2 imprisons the cGAS-STING machinery to abrogate antitumor immunity. Mol Cell. 2021;81:4147–64.e7.
Wang C, Duan Y, Duan G, Wang Q, Zhang K, Deng X, et al. Stress induces dynamic, cytotoxicity-antagonizing TDP-43 nuclear bodies via paraspeckle LncRNA NEAT1-mediated liquid-liquid phase separation. Mol Cell. 2020;79:443–58.e7.
Zong Y, Zhang CS, Li M, Wang W, Wang Z, Hawley SA, et al. Hierarchical activation of compartmentalized pools of AMPK depends on severity of nutrient or energy stress. Cell Res. 2019;29:460–73.
Zhang G, Wang Z, Du Z, Zhang H. mTOR regulates phase separation of PGL granules to modulate their autophagic degradation. Cell. 2018;174:1492–506.e22.
Zhao J, Hu Y, Li H, Liu C, Nie Z, Chen Z, et al. Liquid-liquid phase separation-mediated cellular-scale compartmentalization of hydrogel covalent cross-linking promotes microtubule-based mechanosensing. J Am Chem Soc. 2025;147:14336–47.
Shin Y, Chang YC, Lee DSW, Berry J, Sanders DW, Ronceray P, et al. Liquid nuclear condensates mechanically sense and restructure the genome. Cell. 2018;175:1481–91.e13.
Li C, Hao B, Yang H, Wang K, Fan L, Xiao W. Protein aggregation and biomolecular condensation in hypoxic environments (Review). Int J Mol Med. 2024;53:33.
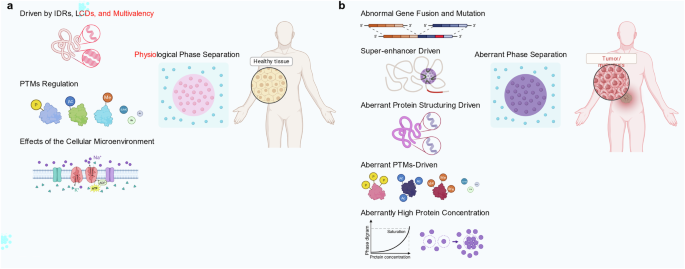
Hu L, Huang Z, Liu Z, Zhang Y. Biomolecular phase separation in tumorigenesis: from aberrant condensates to therapeutic vulnerabilities. Mol Cancer. 2025;24:220.
Shao Y, Zhang Y, Zhu W, Lu H. Biomolecular condensates in Hippo pathway regulation. J Zhejiang Univ Sci B. 2025;26:949–60.
Boija A, Klein IA, Sabari BR, Dall’Agnese A, Coffey EL, Zamudio AV, et al. Transcription factors activate genes through the phase-separation capacity of their activation domains. Cell. 2018;175:1842–55.e16.
Lu B, Zou C, Yang M, He Y, He J, Zhang C, et al. Pharmacological inhibition of core regulatory circuitry liquid-liquid phase separation suppresses metastasis and chemoresistance in osteosarcoma. Adv Sci (Weinh). 2021;8:e2101895.
Zhang C, He Y, Zhan X, Yu J, Xu S, Zhang Q, et al. Defining heterogeneity in core regulatory circuitry reveals HOXB3 condensation as a potential target in glioblastoma. Nat Cell Biol. 2025;27:1848–62.
Xie CC, Wang T, Liu XR, Wang Y, Dang Q, Ding T, et al. Liquid-liquid phase separation in major hallmarks of cancer. Cell Prolif. 2025;0:e70122.
Chen Y, Ying Y, Ma W, Ma H, Shi L, Gao X, et al. Targeting the epigenetic reader ENL inhibits super-enhancer-driven oncogenic transcription and synergizes with BET inhibition to suppress tumor progression. Cancer Res. 2024;84:1237–51.
Li X, Zhu R, Jiang H, Yin Q, Gu J, Chen J, et al. Autophagy enhanced by curcumin ameliorates inflammation in atherogenesis via the TFEB-P300-BRD4 axis. Acta Pharm Sin B. 2022;12:2280–99.
Tran C, Ouk S, Clegg NJ, Chen Y, Watson PA, Arora V, et al. Development of a second-generation antiandrogen for treatment of advanced prostate cancer. Science. 2009;324:787–90.
Meyers PA, Federman N, Daw N, Anderson PM, Davis LE, Kim A, et al. Open-label, multicenter, phase I/II, first-in-human trial of TK216: a first-generation EWS::FLI1 fusion protein antagonist in ewing sarcoma. J Clin Oncol. 2024;42:3725–34.
Li T, Jiang HL, Tong YG, Lu JJ. Targeting the Hsp90-Cdc37-client protein interaction to disrupt Hsp90 chaperone machinery. J Hematol Oncol. 2018;11:59.
Dewpoint Therapeutics Announces Nomination of First C-Mod Development Candidate DPTX3186 for Treatment of Wnt-Driven Cancers [Internet]. Boston(MA): Dewpoint Therapeutics; 2024 Oct 28 [cited 2025 Dec 5]. Available from: https://dewpointx.com/dewpoint-therapeutics-announces-nomination-of-first-c-mod-development-candidate-dptx3186-for-treatment-of-wnt-driven-cancers/.
FDA Grants Orphan Drug Designation to Dewpoint Therapeutics’ DPTX3186 for the Treatment of Gastric Cancer [Internet]. [place unknown]: The National Law Review; 2025 Oct 29 [cited 2025 Dec 5]. Available from: https://natlawreview.com/press-releases/fda-grants-orphan-drug-designation-dewpoint-therapeutics-dptx3186-treatment#google_vignette.
Wang Z, Zhang H. Phase separation, transition, and autophagic degradation of proteins in development and pathogenesis. Trends Cell Biol. 2019;29:417–27.
Chen H, Bao Y, Li X, Chen F, Sugimura R, Zeng X, et al. Cell surface engineering by phase-separated coacervates for antibody display and targeted cancer cell therapy. Angew Chem Int Ed Engl. 2024;63:e202410566.
Wu Y, Zhou L, Zou Y, Zhang Y, Zhang M, Xu L, et al. Disrupting the phase separation of KAT8-IRF1 diminishes PD-L1 expression and promotes antitumor immunity. Nat Cancer. 2023;4:382–400.
Gonzalez-Martinez D, Roth L, Mumford TR, Guan J, Le A, Doebele RC, et al. Oncogenic EML4-ALK assemblies suppress growth factor perception and modulate drug tolerance. Nat Commun. 2024;15:9473.
Scheller K, Zhou X, Li K, Jiang L, Ochoa R, Cossio P, et al. Trimerization domain-interfering peptide inhibits EML4-ALK condensate formation, fusion-dependent signaling, and cell growth. Mol Biol Cell. 2025;36:br25.
Schapira M, Calabrese MF, Bullock AN, Crews CM. Targeted protein degradation: expanding the toolbox. Nat Rev Drug Discov. 2019;18:949–63.
Zhang X, Lee HC, Shirazi F, Baladandayuthapani V, Lin H, Kuiatse I, et al. Protein targeting chimeric molecules specific for bromodomain and extra-terminal motif family proteins are active against pre-clinical models of multiple myeloma. Leukemia. 2018;32:2224–39.
PROTAC shrinks mutated prostate tumors. Cancer Discov. 2022;12:Of2.
Zhao L, Zhao J, Zhong K, Tong A, Jia D. Targeted protein degradation: mechanisms, strategies and application. Signal Transduct Target Ther. 2022;7:113.
Wang S, Huo X, Yang Y, Mo Y, Kollipara RK, Kittler R. Ablation of EWS-FLI1 by USP9X inhibition suppresses cancer cell growth in Ewing sarcoma. Cancer Lett. 2023;552:215984.
Czapiewski R, Gilbert N. Role of RNA in genome folding: It’s all about affinity. Curr Opin Struct Biol. 2025;94:103136.
Mondala PK, Vora AA, Zhou T, Lazzari E, Ladel L, Luo X, et al. Selective antisense oligonucleotide inhibition of human IRF4 prevents malignant myeloma regeneration via cell cycle disruption. Cell Stem Cell. 2021;28:623–36.e9.
Yan X, Yang Z, Cao X, Liang L, Duan Y, Zhang P, et al. Targeting endothelial MYC using siRNA or miR-218 nanoparticles sensitizes chemo- and immuno-therapies by recapitulating the Notch activation-induced tumor vessel normalization. Theranostics. 2025;15:5381–401.
Longoria-García S, Sánchez-Domínguez CN, Sánchez-Domínguez M, Delgado-Balderas JR, Islas-Cisneros JF, Vidal-Gutiérrez O, et al. Design and Characterization of pMyc/pMax peptide-coupled gold nanosystems for targeting Myc in prostate cancer cell lines. Nanomaterials (Basel). 2023;13:2802.
Dall’Agnese G, Hannett NM, Overholt KJ, Platt JM, Henninger JE, Marcos-Vidal A, et al. APE1 condensation in nucleoli of non-cancer cells depends on rRNA transcription and forming G-quadruplex RNA structures. Nucleic Acids Res. 2025;53:gkaf168.
Liu H, Huang L, Luo J, Chen W, Zhang Z, Liao X, et al. Prediction and identification of B cell epitopes derived from EWS/FLI-l fusion protein of Ewing’s sarcoma. Med Oncol. 2012;29:3421–30.
Meyer-Wentrup F, Richter G, Burdach S. Identification of an immunogenic EWS-FLI1-derived HLA-DR-restricted T helper cell epitope. Pediatr Hematol Oncol. 2005;22:297–308.
Gambacorti-Passerini C, Grignani F, Arienti F, Pandolfi PP, Pelicci PG, Parmiani G. Human CD4 lymphocytes specifically recognize a peptide representing the fusion region of the hybrid protein pml/RAR alpha present in acute promyelocytic leukemia cells. Blood. 1993;81:1369–75.
Jia W, Li W, Li Z, Li P. An all-in-one targeted protein degradation platform guided by degradation condensates-bridging bi-specific nanobodies. Cell Res. 2024;34:389–92.
Deng R, Zuo C, Li Y, Xue B, Xun Z, Guo Y, et al. The innate immune effector ISG12a promotes cancer immunity by suppressing the canonical Wnt/β-catenin signaling pathway. Cell Mol Immunol. 2020;17:1163–79.
Pokatayev V, Yang K, Tu X, Dobbs N, Wu J, Kalb RG, et al. Homeostatic regulation of STING protein at the resting state by stabilizer TOLLIP. Nat Immunol. 2020;21:158–67.
Eldesouky LM, El-Moslemany RM, Ramadan AA, Mehanna RA, Kolaib NM, El-Kamel AH. Lactoferrin functionalized bovine serum albumin nanoparticles for synergistic chemo-gene therapy of lung cancer: Co-delivery of tetrandrine and miRNA-126. Int J Biol Macromol. 2025;330:148256.
Younis MA, Sato Y, Elewa YHA, Harashima H. Harnessing the composition of lipid nanoparticles to selectively deliver mRNA to splenic immune cells for anticancer vaccination. Drug Deliv Transl Res. 2025;15:3626–41.
Jiang Z, Li J, Liu G, Qiu Q, Zhang J, Hao M, et al. A pH-sensitive glucose oxidase and hemin coordination micelle for multi-enzyme cascade and amplified cancer chemodynamic therapy. Small. 2024;20:e2407674.
Fan K, Jia X, Zhou M, Wang K, Conde J, He J, et al. Ferritin nanocarrier traverses the blood brain barrier and kills glioma. ACS Nano. 2018;12:4105–15.
Gabathuler R. Approaches to transport therapeutic drugs across the blood-brain barrier to treat brain diseases. Neurobiol Dis. 2010;37:48–57.
Tong F, Hu H, Xu Y, Zhou Y, Xie R, Lei T, et al. Hollow copper sulfide nanoparticles carrying ISRIB for the sensitized photothermal therapy of breast cancer and brain metastases through inhibiting stress granule formation and reprogramming tumor-associated macrophages. Acta Pharm Sin B. 2023;13:3471–88.
Platzbecker U, Avvisati G, Cicconi L, Thiede C, Paoloni F, Vignetti M, et al. Improved outcomes with retinoic acid and arsenic trioxide compared with retinoic acid and chemotherapy in non-high-risk acute promyelocytic leukemia: final results of the randomized Italian-German APL0406 trial. J Clin Oncol. 2017;35:605–12.
Nadiminti KVG, Sahasrabudhe KD, Liu H. Menin inhibitors for the treatment of acute myeloid leukemia: challenges and opportunities ahead. J Hematol Oncol. 2024;17:113.
Issa GC, Aldoss I, Thirman MJ, DiPersio J, Arellano M, Blachly JS, et al. Menin inhibition with revumenib for KMT2A-rearranged relapsed or refractory acute leukemia (AUGMENT-101). J Clin Oncol. 2025;43:75–84.
Arellano ML, Thirman MJ, DiPersio JF, Heiblig M, Stein EM, Schuh AC, et al. Menin inhibition with revumenib for NPM1-mutated relapsed or refractory acute myeloid leukemia: the AUGMENT-101 study. Blood. 2025;146:1065–77.
Wang ES, Montesinos P, Foran J, Erba H, Rodríguez-Arbolí E, Fedorov K, et al. Ziftomenib in relapsed or refractory NPM1-mutated AML. J Clin Oncol. 2025;43:3381–90.
Snyder LB, Neklesa TK, Willard RR, Gordon DA, Pizzano J, Vitale N, et al. Preclinical evaluation of bavdegalutamide (ARV-110), a novel proteolysis targeting chimera androgen receptor degrader. Mol Cancer Ther. 2025;24:511–22.
Campone M, De Laurentiis M, Jhaveri K, Hu X, Ladoire S, Patsouris A, et al. VepdegESTRANT, A PROTAC estrogen receptor degrader, in advanced breast cancer. N Engl J Med. 2025;393:556–68.
Brooks TA, Hurley LH. Targeting MYC expression through G-quadruplexes. Genes Cancer. 2010;1:641–9.
Chen BJ, Wu YL, Tanaka Y, Zhang W. Small molecules targeting c-Myc oncogene: promising anti-cancer therapeutics. Int J Biol Sci. 2014;10:1084–96.
Reilley MJ, McCoon P, Cook C, Lyne P, Kurzrock R, Kim Y, et al. STAT3 antisense oligonucleotide AZD9150 in a subset of patients with heavily pretreated lymphoma: results of a phase 1b trial. J Immunother Cancer. 2018;6:119.
Burnett AK, Russell NH, Hills RK, Bowen D, Kell J, Knapper S, et al. Arsenic trioxide and all-trans retinoic acid treatment for acute promyelocytic leukaemia in all risk groups (AML17): results of a randomised, controlled, phase 3 trial. Lancet Oncol. 2015;16:1295–305.
Esposito V, Benigno D, Bello I, Panza E, Bucci M, Virgilio A, et al. Structural and biological features of G-quadruplex aptamers as promising inhibitors of the STAT3 signaling pathway. Int J Mol Sci. 2023;24:9524.
Noguera NI, Catalano G, Banella C, Divona M, Faraoni I, Ottone T, et al. Acute promyelocytic leukemia: update on the mechanisms of leukemogenesis, resistance and on innovative treatment strategies. Cancers (Basel). 2019;11:1591.
Mancarella C, Morrione A, Scotlandi K. PROTAC-based protein degradation as a promising strategy for targeted therapy in sarcomas. Int J Mol Sci. 2023;24:16346.
Xu H, Hurley LH. A first-in-class clinical G-quadruplex-targeting drug. The bench-to-bedside translation of the fluoroquinolone QQ58 to CX-5461 (Pidnarulex). Bioorg Med Chem Lett. 2022;77:129016.
Alberti S, Arosio P, Best RB, Boeynaems S, Cai D, Collepardo-Guevara R, et al. Current practices in the study of biomolecular condensates: a community comment. Nat Commun. 2025;16:7730.
Steffen CL, Manoharan GB, Pavic K, Yeste-Vázquez A, Knuuttila M, Arora N, et al. Identification of an H-Ras nanocluster disrupting peptide. Commun Biol. 2024;7:837.
Yang X, Wang H, Xie E, Tang B, Mu Q, Song Z, et al. Rewiring ERBB3 and ERK signaling confers resistance to FGFR1 inhibition in gastrointestinal cancer harbored an ERBB3-E928G mutation. Protein Cell. 2020;11:915–20.
Boulay G, Sandoval GJ, Riggi N, Iyer S, Buisson R, Naigles B, et al. Cancer-specific retargeting of BAF complexes by a prion-like domain. Cell. 2017;171:163–78.e19.