Aging Immune Cells Linked to Fatty Liver Disease
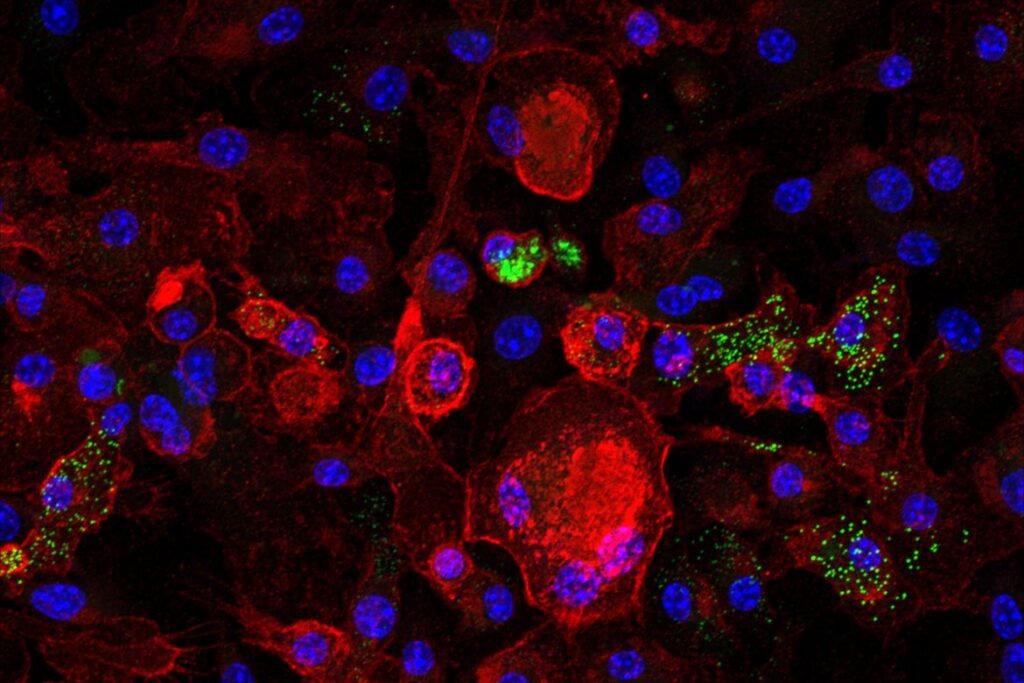

UCLA researchers have found that macrophages with a senescent phenotype may be actively driving progression of fatty liver disease. A study published today in Nature Aging reports that clearing this aging cell population in mice dramatically reduced liver inflammation and reversed damage even without any dietary changes.
“Senescent cells are fairly rare, but think of them like a broken-down car,” said Anthony J. Covarrubias, PhD, assistant professor at the UCLA David Geffen School of Medicine and senior author of the study. “Just one stalled car can back up traffic for miles. Now imagine five or ten of them slowly accumulating. That’s what these cells do to a tissue: even a small number causes enormous disruption.”
As cells age and become senescent, they are known to drive chronic inflammation across a range of tissues. While previous research has shown that eliminating senescent cells can improve health and lengthen lifespan, there is still a limited understanding of which cells undergo the senescence process and how to distinguish them from healthy cells.
This is especially the case for cells that naturally share hallmark features with senescent cells, as is the case of macrophages; when activated, macrophages secrete a range of inflammatory cytokines and immunomodulatory metabolites that many senescent cells also produce when driving chronic, age-driven inflammation.
The researchers found that no single biomarker was enough to identify senescent macrophages. Instead, they identified that this aging cell population was defined by the simultaneous expression of p21 and Trem2 proteins, together with altered nuclear morphology, lipid metabolism and type I interferon (IFN) hyperactivation.
In mice, senescent macrophages carrying this molecular signature were found to surge from 5% in young mice to up to 80% in older ones, correlating with a rise in chronic liver inflammation during normal aging. In addition, excess cholesterol was found to push macrophages into a senescent state where they stopped dividing, increased secretion of inflammatory proteins and activated expression of p21 and Trem2.
“Physiologically, macrophages can handle cholesterol metabolism,” said Ivan A. Salladay-Perez, graduate student in the Covarrubias lab and lead author of the study. “But in a chronic state, it’s pathological. When you look at fatty liver disease, which is driven by overnutrition and too much cholesterol in the blood, that excess cholesterol appears to be a major driver of the senescent macrophage population.”
Experiments using a publicly available genomic dataset of patient liver biopsies found that the same senescent macrophage signature was increased in diseased livers compared to healthy ones, suggesting they also play a role in chronic liver disease in humans.
In mice with a high-fat, high-cholesterol diet, a drug that selectively kills senescent cells was found to reduce overall body weight and make livers healthier—smaller and with a lower fat percentage. These findings suggest that clearing senescent macrophages from the liver does not just slow the progression of fatty liver disease, but can actually reverse it without changing the diet.
Because the drug tested in mice is too toxic for humans, the researchers plan to begin drug screening studies to identify new compounds that can replicate these effects. They will also be exploring whether this therapeutic target could be expanded to a range of other age- and cholesterol-driven conditions where senescent macrophages have been observed.
“It all goes back to understanding how these cells arise in the first place,” said Salladay-Perez. “If you really understand the basic mechanisms driving inflammation with aging, you can target those same mechanisms to treat not just fatty liver disease, but atherosclerosis, Alzheimer’s and cancer.”
